COVID in Context: How does testing for COVID work and what could go wrong?

Welcome to COVID in Context, where staff at The Nevada Independent provide context and clarity on what's happening with COVID-19 in Nevada.
COVID in Context will change depending on the situation at hand. If you have any questions, thoughts or suggestions, please reach out to [email protected], send me a direct message on Twitter, or submit them using this form.
For daily updates, check out our constantly updating data page and follow @nvindycovidbot on Twitter.
The science of lab testing and where there's room for human error
A ProPublica investigation published last month raised eyebrows after it detailed how Northshore Clinical Labs, a COVID testing company with political connections in Nevada, provided faulty tests.
Since then, we've received reader questions about how testing works and what may have produced inaccurate results.
To understand the nuances of the testing and lab certification process, I sat down with Nevada State Public Health Lab Director Mark Pandori on Wednesday. Pandori touched on recommendations for storing at-home COVID tests (in your refrigerator!) and the different processes related to the various testing methods.
The Northshore Labs tests were providing false-negative results — or, in other words, were not detecting the virus, even though someone was infected.
"Some kind of human error is always the most likely thing," Pandori said. "There's a lot of chemicals that you mix together to get a sensitive PCR to run. And if you're getting a false negative … they're either using rotten chemicals, older chemicals, or they're not putting the recipe together correctly."
The following interview has been paraphrased and edited for length and clarity.
What are rapid antigen tests and PCR tests? How do they work?
Antigen tests, frequently referred to as rapid tests, detect protein fragments specific to the coronavirus. After a swab of liquid is taken from the nose and used in the test, the liquid binds to chemicals on the test. Once the liquid binds, a chemical reaction takes place that produces a line indicating whether fragments of the virus are present or not.
On the other hand, a PCR test looks for a specific genomic sequence that is unique to SARS-CoV-2. The PCR test is considered the gold standard in detecting the virus. It is engineered to detect the RNA (or genetic material) specific to the virus and can detect the virus within days of infection, even for those with no symptoms.
What is different between them?
Inherently, tests that look for protein are less sensitive than tests for chromosomes or genomes. PCR tests look for genetic material which is unique to the virus.
When running an antigen test, the chemicals on the strip are antibodies detecting proteins from the virus. Antibodies are inherently less specific because they're protein interactions and other proteins in the human body could bind with the antibodies and produce an incorrect test result.
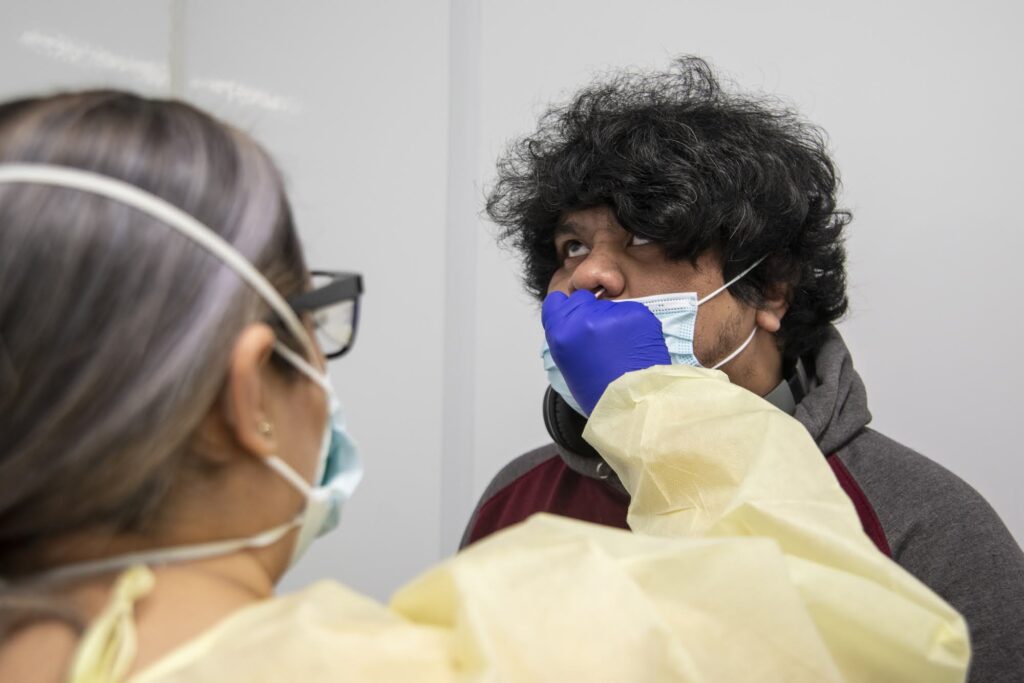
How do health care professionals collect and prep a specimen for testing? What can go wrong during the process?
Like the rapid test, a swab goes into the nose and then is placed into a tube with fluid in it that stabilizes the sample. Once the sample gets to the lab, researchers can test it to see if it contains the virus.
What can go wrong? At the point of collection, if someone is doing it quickly, they may not get a good specimen. If it's a lousy system, someone may mix up two samples. They could also collect the sample correctly but jeopardize the integrity of the result by leaving it in the hot sun too long or not getting it to a lab in a reasonable amount of time.
These tests are biochemicals, and just like all the biochemical junk in your refrigerator, it rots over time, and it rots quicker when it's not cold.
What is the shelf life of a sample after it has been collected?
Using the liquid storage methods, the sample can be out at room temperature for probably eight to 24 hours. But then it needs to be refrigerated, preferably within eight hours. Afterward, it can be at a refrigerated temperature for 72 hours. Then it either needs to be tested, or it has to be frozen at minus 80 degrees Fahrenheit. And once frozen, it's pretty much indefinite.
What can go wrong with an at-home rapid test? What should people pay attention to if they have rapid tests at home?
I pay attention to the expiration date. These tests have chemicals and antibodies on them and proteins. Just like anything else sitting at room temperature, they will decay. And if they decompose, they will lose capability, specificity and sensitivity.
If you've been keeping it stored at an extremely warm temperature, I would be concerned because that would hasten the decay of the chemicals on those strips.
What is the process for looking to get a test authorized?
Right now, the Food and Drug Administration would need to look at the data collected during the research phase. Once you get enough data to compare to an existing test, you would give it to the FDA. When the FDA deems there is enough data available to support efficacy, functionality and safety, officials may authorize emergency use.
When that emergency use authorization goes away, the normal process for getting a test approved will occur.
What does a pre-licensing lab inspection look like and how does a lab get authorized?
How a lab gets authorized is different. You have to show that you've got safety measures, quality assurance and quality management control. You must show that you've hired licensed people. And then you've got to show that you have procedures and validated tests.
Even if a test is approved or cleared in some manner by the FDA, labs need to verify they can run the test. This means that they take that test and run that test at their facilities to demonstrate its validity.
What quality control measures are in place for testing?
So let's say that you are operating a licensed lab and running a COVID PCR test. Once you establish that, a licensed lab must participate in proficiency testing three times a year.
As part of the proficiency test, labs receive five specimens, three times a year from an external organization. The lab must then test those specimens and have at least an 80 percent successful identification rate to pass. If the lab gets lower than 80 percent, it fails the test. If it fails two of the three tests, it cannot run that test anymore.
What happens if you fail?
If a lab fails, it must start from scratch, figure out what went wrong, revalidate a new test, and go through an investigation. So once labs are up and running, if they're not getting 80 percent of tests correct, they're in a lot of trouble.
What are some of the common reasons a lab might get an incorrect result even after passing licensing requirements?
Generally, labs are the ones that are best at keeping specimens at correct temperatures. That's rarely a reason for failure in a lab.
Labs are working with 96 well plates that are 5 inches by 3 inches in size. When technicians are pipetting into those wells, if any splashing occurs and they don't notice, that could cause a false positive reaction. Too often, lab specimens get switched because they're processing thousands in a row. Those would be your most common errors leading to an incorrect result.
We don't see that the test itself fails very often. Usually, it would be caused by an operator error.
— Tabitha Mueller
Understanding the numbers
The number of new COVID-19 cases reported on average each day is on the rise from last week.
As of Monday, 828 new cases were reported on average each day over the last two weeks, up from 673 one week prior – a 23 percent increase. That number is significantly less than the highest point of the winter Omicron surge, 6,410, which the state hit on Jan. 24.
The latest wastewater sampling data from the Southern Nevada Water Authority show increased concentrations of the virus from sites across unincorporated Clark County, Las Vegas, North Las Vegas, Henderson and Boulder City. Overall, the concentration levels in Southern Nevada are lower than those seen at the peak of the Omicron surge in January but are creeping upward.
Concentrations of the virus in wastewater sampling in Washoe County show increases as well.
— Tabitha Mueller
Good news!
In May, The Nevada Independent reported on 13-year-old Bennett Gilles' experience with long COVID and the fatigue he grappled with after he caught the virus in September.
His mother, Alyson Gilles, emailed recently with an update about how he has been feeling:
"He has slowly been getting better, day by day, and a few weeks ago, he seemed to really turn a corner, energy-wise. He set a goal to try out for a competitive soccer team and last night he learned he not only made it, but he made it to the team's premier level! We are hopeful that this is finally behind him! Thank you for sharing his story! The people who reached out after reading your article gave us so much hope that this day would finally arrive."
News you can use
Proof of vaccination
If you got your vaccine in Nevada, you can now access and download your COVID-19 vaccination records through the state's immunization information system here.
If you receive an error message as you try to sign into the system, call 1-800-401-0946 or email [email protected].
Searching for COVID-19 testing and treatment options?
The Nevada Hospital Association has reported that there are free therapies available throughout Nevada for COVID-19 patients, including Evusheld, Sotrovimab, Paxlovid, Molnupiavir, and soon Bebtelovimab.
Patients can complete the pre-screening tool at https://nevadacovidtreatment.com/ or call 800-401-0946 (7 a.m. - 8 p.m. seven days a week) to locate and schedule these treatments.
For those looking for testing, individuals should visit testing sites, not hospital emergency departments.
To find a testing site in Nevada, including pharmacies, head on over to the state's COVID-19 test site finder.
Rapid Tests
Here's the link to order free at-home COVID-19 tests through the United States Postal Service.
As part of a federal effort, private insurance companies will cover at-home test kits purchased at pharmacies. Companies will cover up to eight kits per month, depending on the number of people on a health plan.
Here's a guide for getting your over-the-counter COVID-19 test reimbursed.
Updated COVID-19 Guidelines
The Centers for Disease Control and Prevention updated its COVID-19 guidelines, relying less on new cases to determine risk levels and instead focusing on a combination of three metrics: new COVID-related hospital admissions, percentage of hospital beds occupied by COVID-19 patients and new cases per 100,000 people.
The community levels of COVID-19 are broken into low, medium and high categories.
Recommendations for counties falling within the categories are as follows:
- Low: Stay up-to-date with COVID-19 vaccines and get tested if you have symptoms
- Medium: For those at high risk for severe illness discuss precautions, talk with a health care provider, get tested if you have symptoms and stay up-to-date on COVID-19 vaccinations.
- High: Wear a mask in indoor public places, stay up-to-date with COVID-19 vaccines and get tested if you have symptoms.
As of Thursday, under the new guidelines, Washoe County and Clark County fall under the "medium" COVID-19 community risk level category. All other counties within the state fall within the "low" risk level category.
For complete guidelines and recommendations surrounding quarantine and isolation guidelines, head over to the CDC's COVID-19 Quarantine and Isolation page.
What we're reading
Politico reports that COVID-19 vaccinations for children under the age of 5 are expected to begin on June 21.
How often can you be infected with the coronavirus? The New York Times takes a stab at answering the question in this fascinating article.
"After more than two years writing about this virus it was bizarre finally to be up close and personal with it," San Francisco Chronicle's Erin Allday wrote while reflecting on her personal experience after catching the virus.
The U.S. is in the midst of a late-season rise in flu cases, according to data from the Centers for Disease Control and Prevention. Though experts are unsure why cases of the flu are increasing late in the season, some say the end of mask mandates may play a role.
Support Independent Journalism in Nevada
You’ve enjoyed unlimited access to our reporting because we’re committed to providing independent, accessible journalism for all Nevadans.
But sustaining this work — informing communities, holding leaders accountable, and strengthening civic life — depends on readers like you.
Nevada needs strong, independent journalism. Will you join us?
A gift of any amount helps keep our reporting free and accessible to everyone across our state.
Choose an amount or learn more about membership
