Why we went to court vs. the state of Nevada
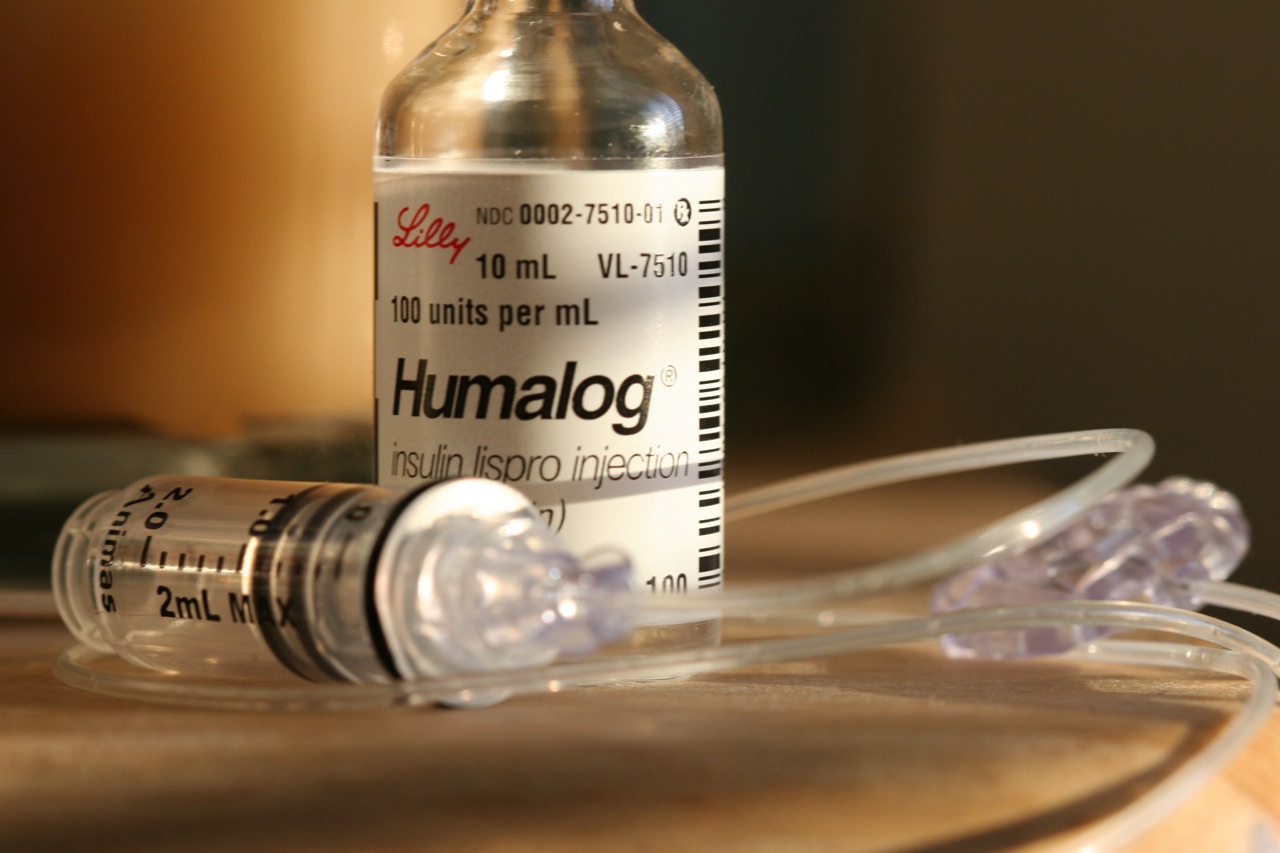
We didn't want to sue. But the state left us no choice.
Today, as Jacob Solis reports, The Nevada Independent went to court to file an action against the state of Nevada for violating Nevada public records statutes by not disclosing information related to a 2017 diabetes drug transparency law. We did not make this decision lightly and have been considering what to do for many months.
We do not like becoming part of any story, but we finally decided the issue is of such overwhelming public interest — drug pricing transparency — that we had to do something about the state's recalcitrance vis a vis a new law designed to make this information less opaque. We believe state officials are flouting the intent of the new law, thus prohibiting patients from knowing what lawmakers wanted them to know.
The intent of the legislation, clearly expressed during the session, was to discover how pharmaceutical companies were pricing insulin in relation to its costs by requiring companies to disclose to the state certain data points associated with the manufacture and sale of the drugs. After much wrangling, including a resurrected measure after a gubernatorial veto, the law passed. But then during the regulatory process, where legislative intent is often left behind, a mechanism was inserted that allows the companies to invoke the federal trade secrets law to essentially undermine what the state did.
To be clear, nobody — legislators, us, the public — wants to expose trade secrets. That's a red herring. Again, the public interest is in how the drugs are priced relative to their costs. That is, are the companies and pharmacy benefit managers (middlemen between the companies and pharmacies), as some legislators believe and anecdotes suggest, gouging people?
Our reporter Megan Messerly, who covered every aspect of this debate and understands the issue better than most, naturally wanted to know what the law had accomplished. She has made two separate public records requests related to the new law — one for the 2018 reports and one for the 2019 reports.
The response from the state was...non-responsive. The Department of Health and Human Services has said it intends to release only what already was available in the public domain such as the drug name, the nonproprietary drug name, the national drug code, the wholesale acquisition cost (list price) and any increase in the list price.
As for the rest of the information the law requires drug companies to disclose to the state, guess how much of the important data the state is willing to release: none.
It's not as if the statute is not clear on what the public is supposed to see by specifically exempting it from the state's trade secret law:
On or before April 1 of each year, the manufacturer of a prescription drug that appears on the most current list compiled by the Department pursuant to subsection 1 of section 3.6 of this act shall prepare and submit to the Department, in the form prescribed by the Department, a report which must include: 1. The costs of producing the drug; 2. The total administrative expenditures relating to the drug, including marketing and advertising costs; 3. The profit that the manufacturer has earned from the drug and the percentage of the manufacturer's total profit for the period during which the manufacturer has marketed the drug for sale that is attributable to the drug; 4. The total amount of financial assistance that the manufacturer has provided through any patient prescription assistance program…
The regulation emanating from the transparency law, though, is what the state — and by extension, the companies — is hanging its hat on. Or, more aptly, using to board up the window of transparency.
It's not surprising that the companies would attempt to get blanket confidentiality when they submitted the required reports. Nor is it surprising they would attempt to cow the state by using the federal Defend Trade Secrets Act — that's why that provision was included in the regulation.
Big Pharma already has sued — and then dropped the action — claiming the 2017 measure is unconstitutional. And the drug companies tried during the most recent session to vitiate the impact, hoping for an amendment to undermine the original bill's intent.
The companies do not want the law to do what it is intended to do — shine a light on how they price drugs, including critical life-saving ones. There is a lot at stake for them. We get that.
But the state should not be an accomplice in the drug companies' efforts to subvert the intent of the landmark law, which has received national attention. Our lawsuit is intended to ensure Carson City abides by the laws that are passed in Carson City, including this important legislation.
(I occasionally will take to this blog to give readers insight into how we made decisions. I welcome your feedback in the comments section or just email me at [email protected].)
Support Independent Journalism in Nevada
You’ve enjoyed unlimited access to our reporting because we’re committed to providing independent, accessible journalism for all Nevadans.
But sustaining this work — informing communities, holding leaders accountable, and strengthening civic life — depends on readers like you.
Nevada needs strong, independent journalism. Will you join us?
A gift of any amount helps keep our reporting free and accessible to everyone across our state.
Choose an amount or learn more about membership
